EMH Schweizerischer Ärzteverlag AG
Farnsburgerstrasse 8
CH-4132 Muttenz
+41 (0)61 467 85 44
support@swisshealthweb.ch
www.swisshealthweb.ch

Publié le 19.07.2022
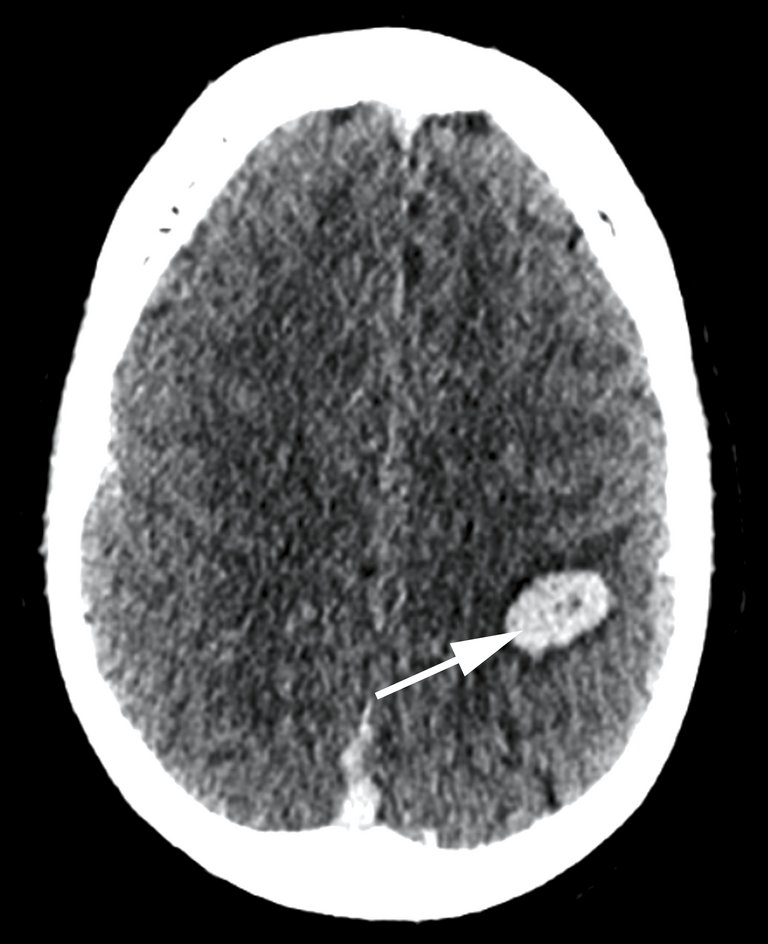
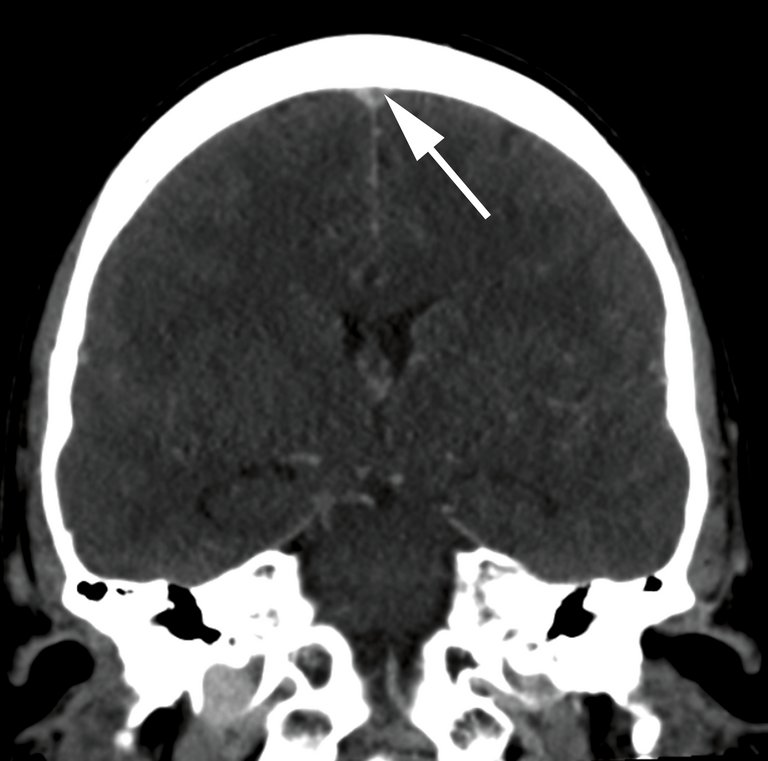
Un homme de 38 ans, jusque-là en bonne santé, s’est présenté en raison d’un trouble de la sensibilité et d’une faiblesse du bras droit nouvellement survenus deux jours auparavant, ainsi que de céphalées du côté droit depuis deux semaines.
| Tableau 1: Facteurs de risque fréquents et causes fréquentes de la thrombose des sinus veineux cérébraux (d’après [2]). | |
| Facteurs de risque/causes | Exemples |
| Thrombophilie | Déficit en antithrombine III |
| Déficit en protéine S | |
| Déficit en protéine C | |
| Résistance à l’APC/facteur V Leiden | |
| Mutation du facteur II G2021 | |
| Anticorps antiphospholipides et anticardiolipines | |
| Hyperhomocystéinémie | |
| Grossesse et post-partum | |
| Contraceptifs oraux | |
| Hormonothérapie de substitution | |
| Tumeurs malignes | Tumeurs cérébrales |
| Hypercoagulabilité dans le cadre de tumeurs en dehors du système nerveux central | |
| Tumeurs malignes hématologiques | |
| Infections | Situées dans le système nerveux central |
| Paraméningées (visage, cou, nez, oreilles, orbite) | |
| Autres localisations | |
| Causes mécaniques | Ponction lombaire |
| Traumatisme crânio-cérébral | |
| Interventions neurochirurgicales | |
| Fermeture d’un cathéter jugulaire | |
| Autres maladies hématologiques | Anémie |
| Polycythémie, thrombocytémie | |
| Maladies systémiques | Maladies de la thyroïde |
| Lupus érythémateux disséminé | |
| Maladie de Behçet | |
| Sarcoïdose | |
| Syndrome néphrotique | |
Publié sous la licence du droit d'auteur.
"Attribution - Non-Commercial - NoDerivatives 4.0"
Pas de réutilisation commerciale sans autorisation..
See: emh.ch/en/emh/rights-and-licences/